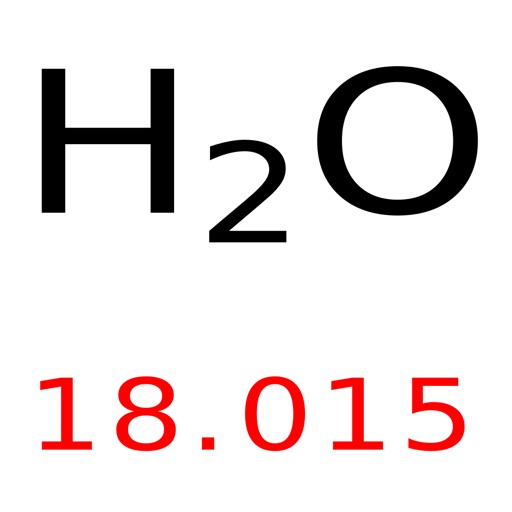
The non-primordial radioisotope 79Se also occurs in minute quantities in uranium ores as a product of nuclear fission. Also naturally occurring is the long-lived primordial radionuclide 82Se, with a half-life of 8.76×10 19 years. Five of these, 74Se, 76Se, 77Se, 78Se, 80Se, are stable, with 80Se being the most abundant (49.6% natural abundance). Selenium has seven naturally occurring isotopes. Selenium does not exhibit the changes in viscosity that sulfur undergoes when gradually heated. With strong reducing agents, it forms polyselenides. It resists oxidation by air and is not attacked by nonoxidizing acids. Unlike the other allotropes, it is insoluble in CS 2. Whereas other selenium forms are insulators, gray selenium is a semiconductor showing appreciable photoconductivity. Gray selenium is formed by mild heating of other allotropes, by slow cooling of molten selenium, or by condensing selenium vapor just below the melting point. The minimum distance between chains is 343.6 pm. The most stable and dense form of selenium is gray and has a chiral hexagonal crystal lattice (space group 152 or 154 depending on the chirality) consisting of helical polymeric chains, where the Se–Se distance is 237.3 pm and Se–Se–Se angle is 103.1°. Other selenium allotropes may contain Se 6 or Se 7 rings. In the Se 8 rings, the Se–Se distance varies depending on where the pair of atoms is in the ring, but the average is 233.5 pm, and the Se–Se–Se angle is on average 105.7°. they are not mapped one onto another by any symmetry operation), and in fact in the γ-monoclinic form, half the rings are in one configuration (and its mirror image) and half in another. The eight atoms of a ring are not equivalent (i.e. They all have a relatively low, monoclinic crystal symmetry ( space group 14) and contain nearly identical puckered cyclooctaselenium (Se 8) rings with different geometric arrangements, as in sulfur. The red α, β, and γ forms are produced from solutions of black selenium by varying the evaporation rate of the solvent (usually CS 2). Upon heating, it softens at 50 ☌ and converts to gray selenium at 180 ☌ the transformation temperature is reduced by presence of halogens and amines. Black selenium is a brittle, lustrous solid that is slightly soluble in CS 2. The structure of black selenium is irregular and complex and consists of polymeric rings with up to 1000 atoms per ring. When rapidly melted, it forms the black, vitreous form, usually sold commercially as beads. When prepared in chemical reactions, selenium is usually an amorphous, brick-red powder. Selenium forms several allotropes that interconvert with temperature changes, depending somewhat on the rate of temperature change. Characteristics Physical properties Structure of hexagonal (gray) selenium Selenium requirements in plants differ by species, with some plants requiring relatively large amounts and others apparently not requiring any. Selenium is listed as an ingredient in many multivitamins and other dietary supplements, as well as in infant formula, and is a component of the antioxidant enzymes glutathione peroxidase and thioredoxin reductase (which indirectly reduce certain oxidized molecules in animals and some plants) as well as in three deiodinase enzymes. 
Selenium is still used in a few types of DC power surge protectors and one type of fluorescent quantum dot.Īlthough trace amounts of selenium are necessary for cellular function in many animals, including humans, both elemental selenium and (especially) selenium salts are toxic in even small doses, causing selenosis. Applications in electronics, once important, have been mostly replaced with silicon semiconductor devices. Selenium is a semiconductor and is used in photocells. The chief commercial uses for selenium today are glassmaking and pigments. Minerals that are pure selenide or selenate compounds are known but rare. Commercially, selenium is produced as a byproduct in the refining of these ores, most often during production. Selenium is found in metal sulfide ores, where it partially replaces the sulfur. Selenium (from Ancient Greek σελήνη ( selḗnē) 'moon') was discovered in 1817 by Jöns Jacob Berzelius, who noted the similarity of the new element to the previously discovered tellurium (named for the Earth). It seldom occurs in its elemental state or as pure ore compounds in Earth's crust. It is a nonmetal (more rarely considered a metalloid) with properties that are intermediate between the elements above and below in the periodic table, sulfur and tellurium, and also has similarities to arsenic. 
Selenium is a chemical element it has the symbol Se and atomic number 34.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
